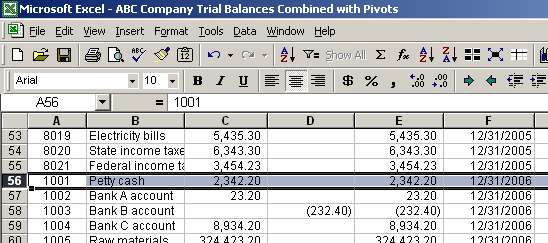
The 90% upper-limit fluxes at a pivot energy of 1 TeV for the source... | Download Scientific Diagram

VALBIOTIS : VALEDIA® : lancement de l'essai clinique « pivot » REVERSE-IT pour l'obtention de l'allégation santé | Business Wire

PDF) PIVOT-12: a Phase III study of adjuvant bempegaldesleukin plus nivolumab in resected stage III/IV melanoma at high risk for recurrence
ENSAYOS CLÍNICOS MELANOMA, TUMORES CUTÁNEOS, SNC, PULMÓN, SARCOMA Y CABEZA CUELLO. C4221016 (STARBOARD) CERRADA FASE SLI, PEN

6 months versus 12 months of adjuvant trastuzumab in early breast cancer (PHARE): final analysis of a multicentre, open-label, phase 3 randomised trial - The Lancet

PTC518 treatment shows benefit for Huntington's in PIVOT-HD trial | Oral therapy safely lowers HTT levels in Phase 2a study: Early data | Huntington's Disease News

A systematic review of interleukin-2-based immunotherapies in clinical trials for cancer and autoimmune diseases - eBioMedicine

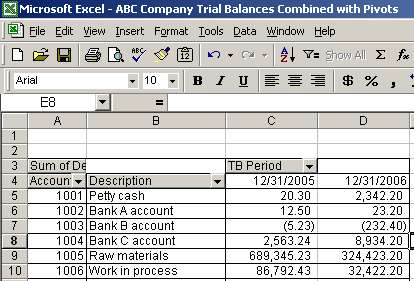
Simulation and Trial & Error Results (Using Formulas & Pivot Table) to... | Download Scientific Diagram

PIVOT-12: a phase III study of adjuvant bempegaldesleukin plus nivolumab in resected stage III/IV melanoma at high risk for recurrence | Future Oncology
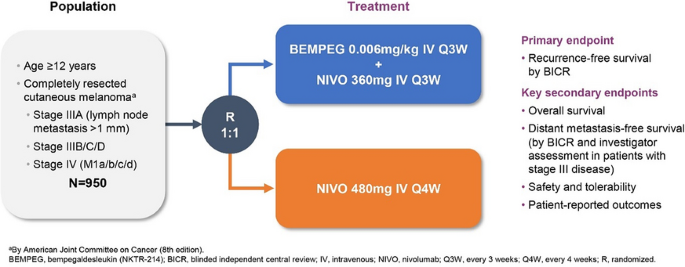
Immunotherapy Bridge 2021 and Melanoma Bridge 2021: meeting abstracts | Journal of Translational Medicine | Full Text

6 versus 12 months of adjuvant trastuzumab for HER2-positive early breast cancer (PERSEPHONE): 4-year disease-free survival results of a randomised phase 3 non-inferiority trial - The Lancet