Purification of active pharmaceutical ingredients (APIS) - Zean Process Engineering. Empresa de ingeniería química

Purification of active pharmaceutical ingredients (APIS) - Zean Process Engineering. Empresa de ingeniería química

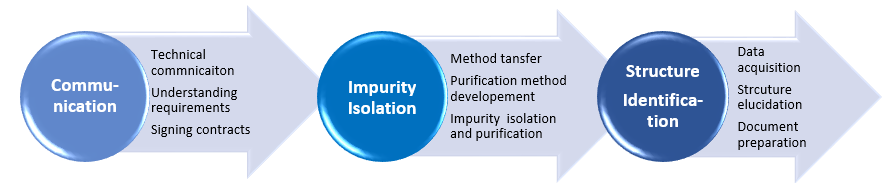
Purification and mass spectrometry study of Maillard reaction impurities in five acyclic nucleoside antiviral drugs - ScienceDirect
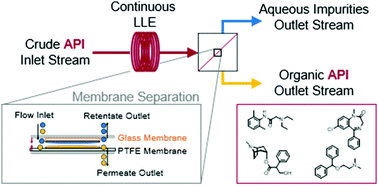
Continuous purification of active pharmaceutical ingredients utilizing polymer membrane surface wettability - Chemical Communications (RSC Publishing)
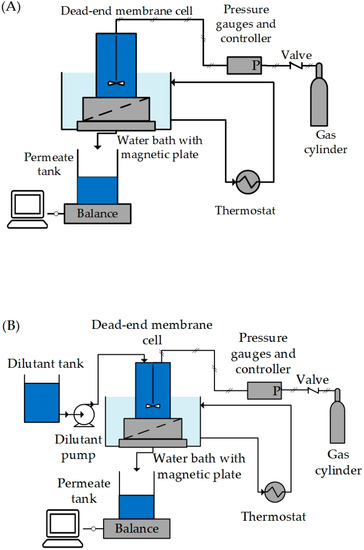
Membranes | Free Full-Text | Membrane-Based Solvent Exchange Process for Purification of API Crystal Suspensions

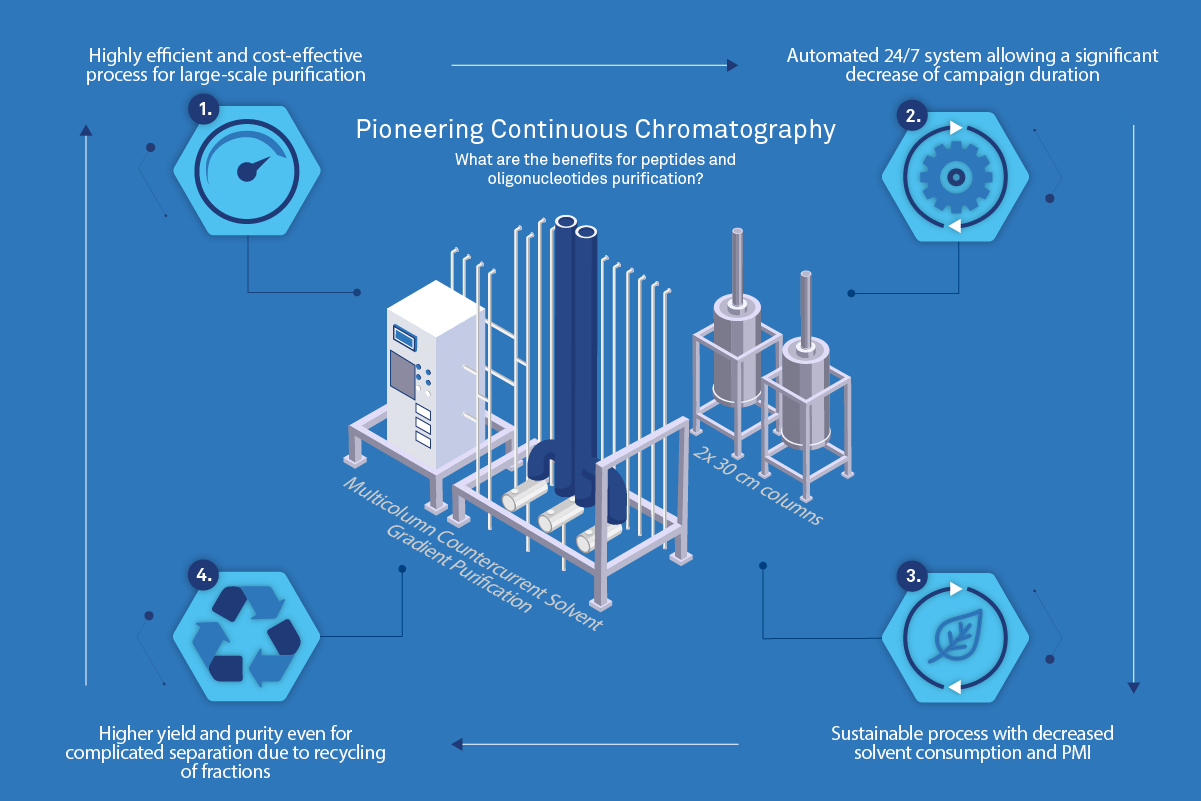
Purification solutions in large-scale manufacturing of oligonucleotide APIs - Chemical Industry Journal

Four module system for the continuous synthesis and purification of... | Download Scientific Diagram

RP-HPLC purification and SDS–PAGE of antimicrobial peptide fractions... | Download Scientific Diagram